This article explores the carbon footprint of cathodic protection deep anode systems and compares it to that of a typical passenger car.
Deep Anode Systems
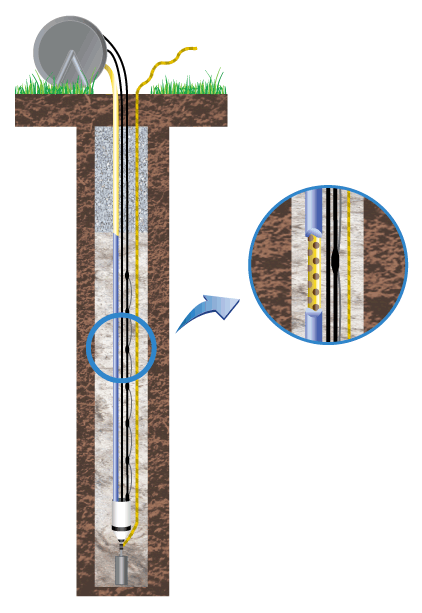
Deep Anode Systems are commonly used throughout the CP industry as a cost-effective means to discharge significant amounts of current to protect pipelines over long distances or large structures in a small area. One of the common components in a deep anode system design is the vent pipe.

The deep anode system vent pipe serves two important related functions:
- To prevent gas blockage that will impede the operation of the anode system
- Prevents the accumulation of chlorine concentrations where chlorides are available
Both issues are directly related to the electro-chemical reactions that occur at the anode to coke backfill, and coke backfill to earth interfaces.
There are two basic types of anodes used in deep anode systems—conventional “massive” anodes, and dimensionally stable anodes.
The conventional “massive” anodes are those anodes that consume as part of the electro-chemical reaction and as such their mass is critical in determining the system’s performance life. The dimensionally stable anodes, typically Mixed Metal Oxide (MMO), are catalytic in nature and do not consume as part of the anodic reaction.
Cathodic Protection Reactions
The primary cathodic protection reactions all involve generating gas:
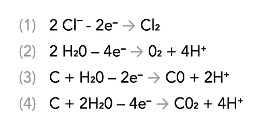
In a properly functioning deep anode system, the gases that occur from these reactions predominantly involve the coke backfill creating carbon monoxide and carbon dioxide. If chlorides are present, some percentage of chlorine gas will also be generated.
The Importance of Venting the Deep Anode System
The gases generated in the coke column typically do not rapidly diffuse into the earth around the coke column and thus will build up. These gases are not electrically conductive and once enough gas builds up around the anode, then the anode can no longer effectively discharge current—a phenomenon known as gas blockage. If Chlorides are present, the chlorine gas reacts with water to create hydrochloric and hypochlorous acids that can attack the cable insulation and cause permanent damage. This is why it is important to properly vent these gases that are a part of the electro-chemical reaction that must occur for CP to function.
What’s the Carbon Footprint?
Given that deep anode systems generate gas, an interesting, although not commonly asked question, is how much carbon dioxide a typical deep anode system generates—in other words, what is the carbon footprint of a deep anode cathodic protection system.
With a few worst-case assumptions and a little stoichiometric chemistry analysis we can answer this question. Assuming all the reactions are generating carbon dioxide and there is no oxygen generation, then for every 4 electrons generated, one CO2 molecule is generated.
One amp-year is equal to 3.1536 x 107 amp seconds or coulombs. One Faraday or 96.487 coulombs is equal to one mole of electrons, therefore, one amp-year is equal to 326.84 moles of electrons. With the 4 to 1 ratio of electrons to CO2, that means that for every mole of electrons, 0.25 moles of CO2 are generated. CO2 has a molar mass of 44.01 g/mol, so for each amp year a total mass of 3,596 grams of CO2 is generated.
For a nominal 50 amp anode system, that would mean a maximum generation of 180 kg of CO2 per year if CO2 was the only gas generated.
How much CO2 is 180 kg/year?
The EPA estimates that the typical passenger vehicle generates 4,600 kg of CO2 per year.
Therefore, your 50 amp deep anode system generates about 4%—or just 1/25th—of what a typical passenger car generates annually.
If you have other technical questions, or for information on MATCOR’s deep anode cathodic protection solutions, please contact us at the link below.






